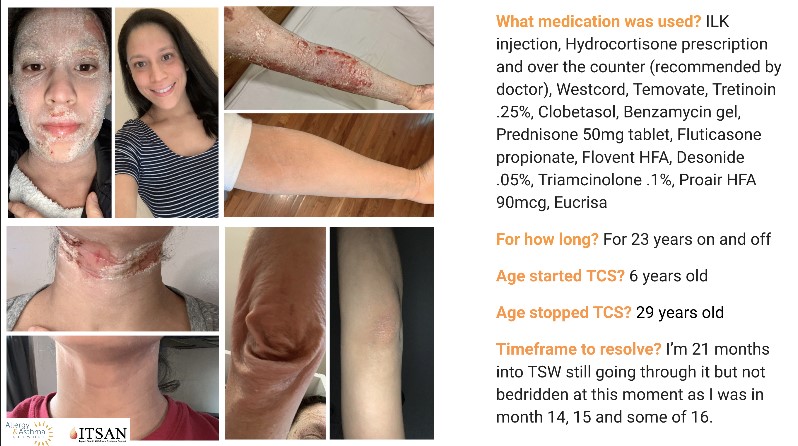
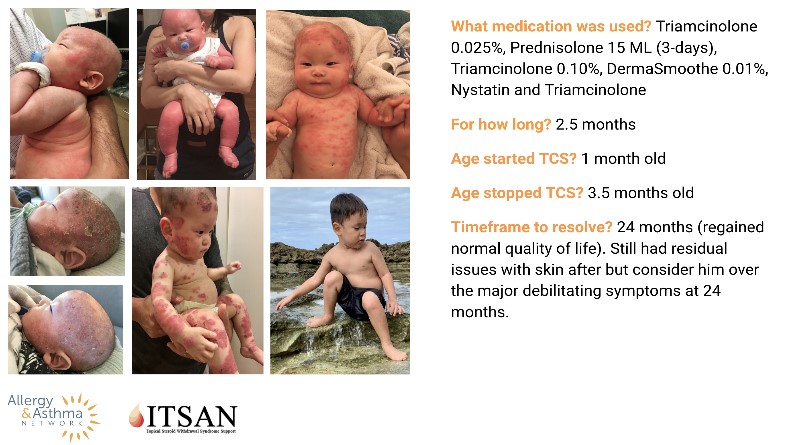
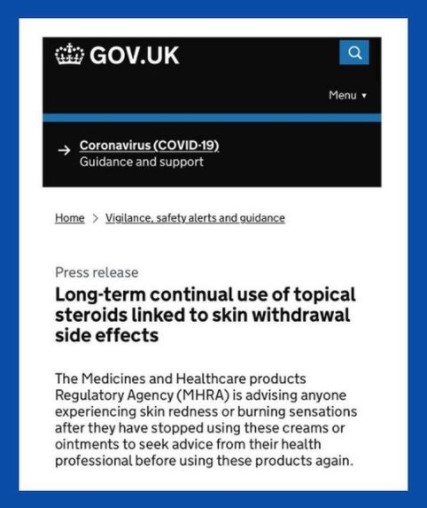
Third Quarter 2021 Newsletter
Hello TSW Community!
We have so much to share about the steps towards raising Topical Steroid Withdrawal Syndrome awareness and prevention that brought us hope recently but before we do, we want to say keep up the hope where you are too. We know that TSW Syndrome affects every part of your life and it takes strength and courage to speak up about this preventable condition, whether to educate a loved one, communicate your needs with your doctor or your child’s doctor, or to reach out for mental health support. We are with you and know that each person has different needs during TSW and everyone deserves to be validated, to access a proper diagnosis and to have support at every healing milestone.
With you in mind, we continue the work of important partnerships that will help improve the lives of TSW sufferers and caregivers. This is a responsibility we think of every day, as we witness the growing need for support and as we advocate for TSW awareness and prevention. In October we reached 11K members in ITSAN’s Private Facebook Support Group, a place where members come together with different, personal TSW healing journeys. ITSAN’s Instagram has more than doubled to 4.7K followers this past year. TSW awareness is spreading and change is coming. In this issue we will highlight ITSAN’s effort to speak up for potentially millions of TSW sufferers worldwide, and how working in partnership with stakeholders in the medical community is leading to change.
ITSAN Partnering for Change
- ITSAN President Kathryn Tullos joined Dr. Peter Lio to present “The Skinny on TSW and Steroids” at the National Eczema Association’s Virtual Eczema Expo on August 28. Discussion included the severe potential side effects of topical steroid use and the need for TSW research, physician and patient education, and safer topical steroid prescribing and monitoring guidelines.
A recent Dermatology Times article, New Frontiers Open for AD, Eczema Solutions, highlighted Eczema Expo discussion: “Diagnostic criteria are lacking, Lio said, but several important features can help identify TSW. The most sensitive signal, he said, is that when patients who have used long-term TCS discontinue treatment, eczema flares up, often at levels worse than its initial presentation.”
- ITSAN President Kathryn Tullos and Executive Assistant Jolene MacDonald joined NEA’s Eczema Expo panel, “TSW: What Now?” with Dr. Eric Simpson, Briana Banos the creator of Preventable and Linette Roungchun, TSW and eczema activist. Discussion included the current struggle many face to access an accurate diagnosis and the importance of recognition of TSW among medical providers, along with shared decision making between doctor and patient.
Recordings of these events are currently only accessible to Expo attendees. We hope to provide access to the recorded sessions when they become available in early January 2022.